화학 합성 공정 제어 시스템 온도 제어 시스템 TCU
Today, let’s talk about our DCS integrated control system (temperature and other control of synthetic process).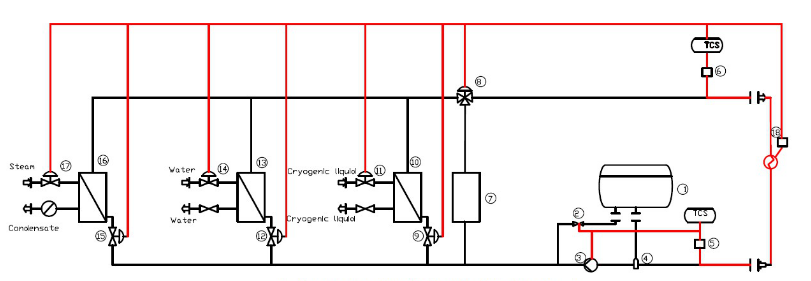
Scalability: the system is an open system, which provides standard TCP / P data communication interface protocol, interface software and application software interface. It has good flexibility and scalability, and meets the requirements of continuously improving production scale for measurement and control ability.
Security: the system has a permission mechanism to realize multi-level permission management. The operating users are divided into five different levels, and the operating permissions of users at different levels are also different. The control system is a relatively closed network, and a firewall is configured between the system network and the enterprise LAN or external network, which can effectively prevent viruses and network attacks and create a safe environment for the operation of the system.
Improve the stability between batches of products: according to the process requirements of pharmaceutical batch production, establish an appropriate production model and strengthen the management of pharmaceutical batches. The batch management of pharmaceutical production is realized through the management modules of production plan designation, production plan release and production plan tracking.
Ensure the integrity of production data: through the application of GMP compliant computer management system and automatic control system, realize the complete automatic collection and monitoring of production data, and carry out effective, accurate and complete data collection, analysis and processing through the computer system, so as to avoid the problems such as lag, missing and error of production data easily caused by the traditional manual recording method.
Improve the traceability of production records: the traceability of pharmaceutical production data is GMP The necessary contents of FDA and other pharmaceutical industry certification, the application of pharmaceutical production automation control system and pharmaceutical intelligent production quality information management system software, real-time, complete and accurate comprehensive records of pharmaceutical production data such as batch, product name, time, production equipment number and operators, and the process of reverse retrieval of production data, which can restore the production status of each production link, Enable producers to analyze the causes of product quality defects, and enable producers to clearly understand the actual production status through the data traceability function, so as to provide data basis for improving production. Technological innovation: realize the real-time monitoring and regulation of key process parameters and quality, clarify the key process control parameters and product characteristics, and then optimize the production process and improve the product quality.
Enhance the market competitiveness. Through the application of advanced pharmaceutical equipment and its automatic control technology, the technical level of Danzhu Dingyan product has greatly improved the product quality and output, and improved the market competitiveness of products.
관련 권장 사항
-
LNEYA 냉각기의 원리
1159산업용 냉각기는 일반적으로 사출 성형, 공구 및 다이 커팅, 식음료, 화학, 레이저, 공작 기계, 반도체 등 다양한 응용 분야의 제품 및 기계를 냉각하는 데 사용됩니다. 대부분의 경우...
세부 정보 보기 -
-
고온 및 저온 액체 순환 냉각기의 얼음 막힘을 해결하기 위한 작은 쿠데타
854In the operation of high and low temperature liquid circulating chillers, ice blockage is a relatively common fault. Based on the experience of relevant manufacturers, we have specially compiled relevant tips for dealing with ice jams. In the pip...
세부 정보 보기 -
2023년 LNEYA 춘절 연휴 안내
1569I have been busy all year, and I am happy to celebrate the new year. The Chinese New Year is coming, and LNEYA is also preparing for the Chinese New Year holiday! Combined with the specific situation of the company, the specific arrangements for ...
세부 정보 보기
 LNEYA 산업용 냉각기 제조업체 공급 업체
LNEYA 산업용 냉각기 제조업체 공급 업체














